 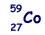
These sub-energy levels are also called orbital. Electron configuration of rhodium through orbitalĪtomic energy shells are subdivided into sub-energy levels. The electron configuration of all the elements can be done through the orbital diagram. The electron configuration of an element with an atomic number greater than 18 cannot be properly determined according to the Bohr atomic model. Electrons can be arranged correctly through orbits from elements 1 to 18. Rhodium shows exceptional electron configuration for equal energy orbitals. Therefore, the order of the number of electrons in each shell of the rhodium atom is 2, 8, 18, 16, 1. Therefore, a rhodium atom will have two electrons in the first shell, eight in the 2nd orbit, and eighteen electrons in the 3rd shell.Īccording to Bohr’s formula, the fourth shell will have seventeen electrons but the fourth shell of rhodium will have sixteen electrons and the remaining one electron will be in the fifth shell. That is, the number of electrons in rhodium is forty-five. Rhodium atom electron configuration (Bohr model) The atomic number is the number of electrons in that element. Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons. The maximum electrons holding capacity in N orbit is 2n 2 = 2 × 4 2 = 32. The maximum electrons holding capacity in M orbit is 2n 2 = 2 × 3 2 = 18. The maximum electron holding capacity in L orbit is 2n 2 = 2 × 2 2 = 8. The maximum electron holding capacity in K orbit is 2n 2 = 2 × 1 2 = 2. The electron holding capacity of each orbit is 2n 2. K is the name of the first orbit, L is the second, M is the third, and N is the name of the fourth orbit. These circular paths are called orbit(shell). The electrons of the atom revolve around the nucleus in a certain circular path. The complete idea of the orbit is given there. Scientist Niels Bohr was the first to give an idea of the atom’s orbit. Rhodium atom electron configuration through orbit For example Aufbau principle, Hund’s principle, and Pauli’s exclusion principle. Electron configuration through orbital (Aufbau principle)Įlectron configuration through orbitals follows different principles.Electron configuration through orbit (Bohr principle).Electron configuration can be done in two ways. The electron configuration of rhodium is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 8 5s 1, if the electron arrangement is through orbitals.  
The arrangement of electrons in rhodium in specific rules in different orbits and orbitals is called the electron configuration of rhodium. These electrons are arranged according to specific rules in different orbitals. The total number of electrons in rhodium is forty-five. What is the electron configuration of rhodium? In this article, I have discussed in detail how to easily write the complete electron configuration of rhodium. The remaining #"7 e"^(-)# will now be distributed in the 3d-subshell.Rhodium is the 45th element in the periodic table and its symbol is ‘Rh’. Because the 4s orbital is filled before the 3d-orbitals, the next two electrons are going to be distributed on the fourth energy level Taking it one subshell at a time, you will have #color(red)(cancel(color(black)(n=3 -> "17 e"^(-))))# in the #3s#, #3p#, and #3d# subshellsĪnd conclude that the electrons that surround the nucleus of a cobalt atom are spread out on #3# energy levels. The third energy level can hold #"18 e"^(-)#, so in theory it can hold the remaining This means that you must fill the 4s-orbital first, then distribute the rest of the electrons to the 3d-orbitals. Now, it's very important to remember that when you're adding electrons to an atom, the 3d-orbitals, which are located on the third energy level, are higher in energy than the 4s-orbital. These electrons will be placed in orbitals in order of increasing energy in accordance to the Aufbau Principle. In your case, cobalt, #"Co"#, is said to have a total of #27# electrons surrounding its nucleus. You can use this equation to find the maximum number of electrons that can be added to each energy level. The relationship that exists between the energy level, #n#, and the number of electrons it can hold can be written like this The number of electrons each energy level can hold increases as you add more and more energy levels to an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
